This study involved four major activities in estimating the historic, current and forecasted size of the antibody therapeutics market. Exhaustive secondary research was carried out to collect information on the market, and its peer markets. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. After that, market breakdown and data triangulation procedures were used to estimate the market size of segments. The revenues for mapped antibody therapeutics products approved till 2023 and clinical pipeline data with estimated launch dates between 2024 and 2027 have been considered for market estimation.
Secondary Research
The secondary sources referred to for this research study include publications from public sources, such as the World Health Organization (WHO), National Institutes of Health (NIH), United States Food and Drug Administration (US FDA), European Medicines Agency (EMA), National Center for Biotechnology Information (NCBI), BioPharm International, ScienceDirect, Purple Book, and clinicaltrial.gov, among others. Secondary sources also include corporate and regulatory filings (such as annual reports, SEC filings, investor presentations, and financial statements); business magazines and research journals; press releases; and trade, business, and professional associations. Secondary data was collected and analyzed to arrive at the overall size of the global antibody therapeutics market, which was validated through primary research.
Primary Research
In-depth interviews were conducted with various primary respondents, including key industry participants, subject-matter experts (SMEs), C-level executives of key market players, and industry consultants, among other experts, to obtain and verify the critical qualitative and quantitative information as well as assess prospects of the market. Various primary sources from both the supply and demand sides of the market were interviewed to obtain qualitative and quantitative information.
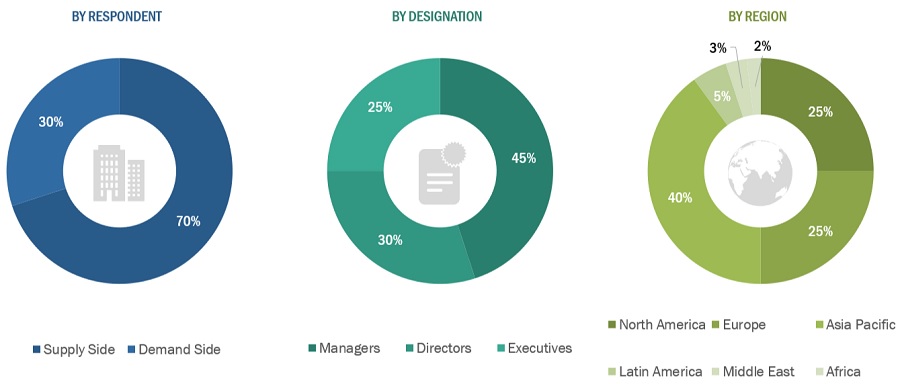
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
Bottom-up approach was used to estimate and validate the total size of the antibody therapeutics market. These methods were also used extensively to estimate the size of various subsegments in the market. The research methodology used to estimate the market size includes the following:
-
The antibody therapeutics products approved by key regulatory authorities so far and which are marketed were mapped along with their revenues from 2020 to 2022 to evaluate the total market size
-
Study about the key industry trends and issues defining the strategic growth objectives of market players was conducted
-
The key players in the industry and market have been identified through extensive secondary research
-
The revenues generated from the antibody therapeutics business of leading players (approved product revenues) have been validated through primary and secondary research
-
All percentage shares, splits, and breakdowns have been determined using secondary sources and verified through primary sources.
Global Antibody Therapeutics Market Size: Bottom-Up Approach

To know about the assumptions considered for the study, Request for Free Sample Report
Data Triangulation
After arriving at the overall market size from the market size estimation process, the total market was split into several segments. To complete the overall market engineering process and arrive at the exact statistics for all segments, data triangulation and market breakdown procedures were employed, wherever applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides.
Market Definition
Therapeutic antibodies are targeted biopharmaceuticals that trigger a biological response. Therapeutic antibodies are drugs that use antibodies which play a significant role in the inherent immune system of the body. When pathogens such as bacteria and viruses invade the body, antibodies are produced from B cells, a type of immune cell, to attack foreign substances (antigens). These antibodies have the characteristics of recognizing and attacking only specific antigens.
Key Stakeholders
-
Pharmaceutical companies
-
Biotechnology companies
-
Research institutions & academic centers
-
Regulatory agencies
-
Health insurers and payers
-
Private & government-funding organizations
Report Objectives
-
To define, describe, and forecast the global antibody therapeutics market by format, disease area, route of administration, source, end user and region
-
To strategically analyze micromarkets1 with respect to individual growth trends, prospects, and contributions to the overall antibody therapeutics market
-
To analyze the opportunities in the market for stakeholders and provide details of the competitive landscape for market leaders
-
To provide detailed information regarding the major factors influencing the market growth, such as drivers, restraints, opportunities, and challenges
-
To forecast the size of the market segments with respect to six main regions: North America, Europe, the Asia Pacific, Latin America, the Middle East, and Africa
-
To strategically analyze the market structure and profile the key players of the global antibody therapeutics market and comprehensively analyze their core competencies
-
To track and analyze competitive developments such as expansions, acquisitions, partnerships, collaborations, agreements, and product launches in the antibody therapeutics market
Available Customizations
With the given market data, MarketsandMarkets offers customizations as per the company’s specific needs. The following customization options are available for this report:
Company Information
-
Twenty five company profiles



Growth opportunities and latent adjacency in Antibody Therapeutics Market