
Next Generation Drug Conjugates (NDCs) Market
Next Generation Drug Conjugates Market by Product (Enhertu, Amvuttra), Type (Antibody-Small Molecule), Target Ligand (Antibody, Peptide), Payload Type (Oligonucleotide, Radionuclide), Indication (Breast Cancer, Prostate Cancer) - Global Forecast to 2035




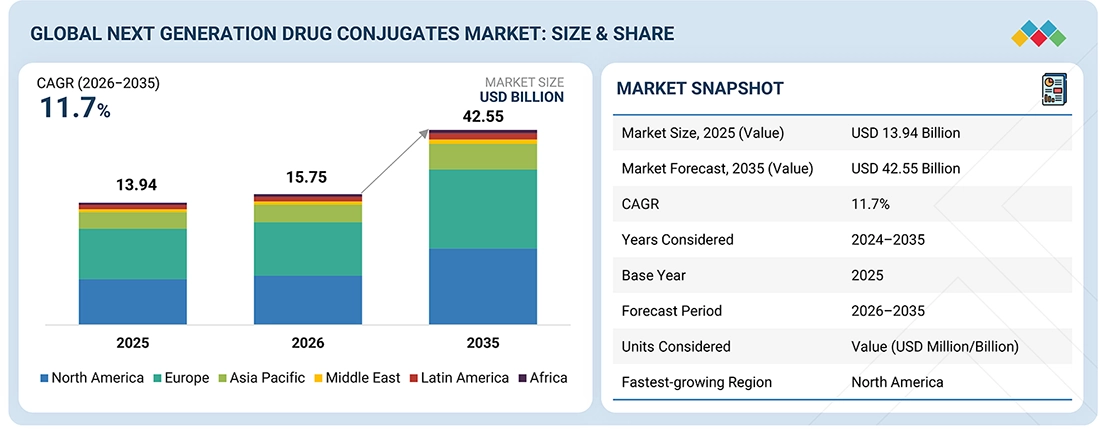
NEXT GENERATION DRUG CONJUGATES MARKET SIZE, SHARE & GROWTH SNAPSHOT
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
The global next generation drug conjugates market is projected to grow from USD 15.75 billion in 2026 to USD 42.55 billion by 2035, at a CAGR of 11.7% during the forecast period. The market was valued at USD 13.94 billion in 2025. Next generation drug conjugates are delivery systems that are biologically or chemically engineered targeting moieties (e.g., antibody, peptide, small molecules, oligonucleotides), a drug (e.g., cytotoxic, radionuclide, nucleic acid), a linker, and conjugation technologies that distinguish next generation conjugates from earlier generations.
KEY TAKEAWAYS
-
BY REGIONNorth America is projected to be the fastest-growing regional segment at a CAGR of 13.2% during the forecast period.
-
BY TYPEBy type, the peptide-radionuclide conjugate is projected to witness the highest CAGR of 14.6% between 2026 and 2035.
-
BY IndicationBy indication, breast cancer is expected to dominate the global next generation drug conjugates market in 2025 with a share of 45.9%.
-
COMPETITIVE LANDSCAPE - Key PlayersAstraZeneca, Daiichi Sankyo, and Novartis were identified as some of the star players in the global next generation drug conjugates market, given their strong market share and product footprint.
-
COMPETITIVE LANDSCAPE - StartupsAvidity Biosciences and Bicycle Therapeutics have distinguished themselves among startups and SMEs by securing strong footholds in specialized niche areas, underscoring their potential as emerging market leaders.
The global next generation drug conjugates (NGDC) market is expanding rapidly, driven by the rising adoption of precision medicine frameworks and the increasing use of biomarker-guided diagnostics across oncology and other complex disease indications.
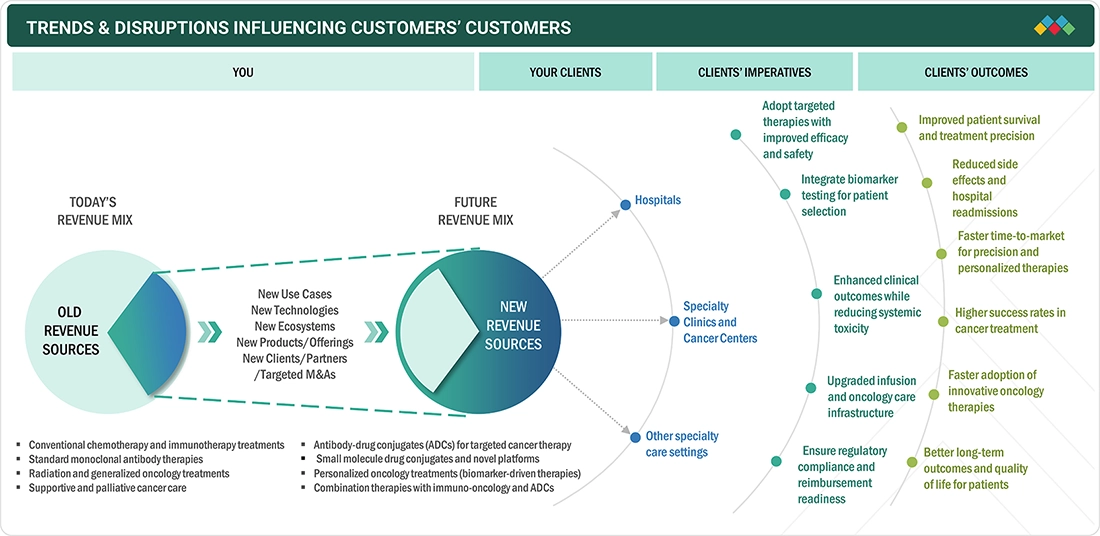
TRENDS & DISRUPTIONS IMPACTING CUSTOMERS' CUSTOMERS
The next generation drug conjugates (NGDC) market is advancing rapidly, driven by innovations in targeting ligands, linker technologies, and payload engineering aligned with precision medicine and targeted biologic therapies. The rising global prevalence of cancer and complex chronic diseases is accelerating demand for highly selective, low-toxicity treatments. Expanding pipelines of antibody-drug conjugates (ADCs), peptide-drug conjugates (PDCs), and other ligand-based platforms are enabling precise delivery of potent therapeutics. Clinically validated, scalable technologies such as site-specific conjugation and advanced linkers are gaining traction, supporting improved outcomes, personalized treatment approaches, and alignment with evolving regulatory and payer expectations across major healthcare markets.
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
MARKET DYNAMICS
Level
-
Increasing clinical success and regulatory approvals of antibody-drug conjugates

-
Technological advancements in linker chemistry and payload development
Level
-
High development and manufacturing costs associated with complex biologics
Level
-
Expansion of drug conjugates into non-oncology therapeutic areas
-
Increasing developments of novel conjugate platforms
Level
-
Complex clinical trial design and patient selection requirements
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Driver: Increasing clinical success and regulatory approvals of antibody-drug conjugates
Increasing clinical success and regulatory approvals of antibody-drug conjugates are key drivers for the NGDC market, validating targeted delivery approaches, improving therapeutic outcomes, and encouraging investment in advanced conjugation technologies, thereby accelerating pipeline expansion and broader adoption across oncology and other disease indications.
Restraint: High development and manufacturing costs associated with complex biologics
High development and manufacturing costs associated with complex biologics act as a restraint, limiting accessibility and scalability, increasing pricing pressures, and posing challenges for smaller players in advancing and commercializing next generation conjugate therapies.
Opportunity: Expansion of drug conjugates into non-oncology therapeutic areas
Expansion of drug conjugates into non-oncology therapeutic areas presents a significant opportunity for the NGDC market, enabling broader clinical applications in cardiovascular, neurological, and autoimmune diseases, while driving innovation in targeted delivery platforms and diversifying revenue streams beyond oncology-focused treatments.
Challenge: Complex clinical trial design and patient selection requirements
Complex clinical trial design and stringent patient selection requirements pose a challenge, as biomarker-driven stratification, smaller target populations, and intricate protocols increase development timelines, operational costs, and regulatory complexity, potentially slowing overall product commercialization.
NEXT GENERATION DRUG CONJUGATES (NDCS) MARKET: COMMERCIAL USE CASES ACROSS INDUSTRIES
| COMPANY | USE CASE DESCRIPTION | BENEFITS |
|---|---|---|
 |
Utilizes its DXd (deruxtecan-based) payload platform in next generation ADCs such as HER2-targeting therapies for breast and gastric cancers | Provides enhanced potency and tumor penetration with improved response rates, addressing resistant and heterogeneous tumor profiles |
 |
Develops pyrrolobenzodiazepine (PBD)-based ADCs targeting hematologic malignancies and expanding into solid tumor indications | Delivers ultra-potent payloads that enable effective treatment of aggressive and refractory cancers, improving patient outcomes in niche oncology segments |
Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
MARKET ECOSYSTEM
The global next generation drug conjugates (NGDC) market is a highly interconnected ecosystem where biopharmaceutical companies, biotechnology firms and research institutions collaborate to develop targeted therapeutic solutions. Drug developers, along with specialized technology providers, enable advanced conjugation platforms integrating antibodies, peptides, and other targeting ligands with potent payloads. Contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) support preclinical development, clinical trials, and commercialization of NGDCs. Regulatory bodies, healthcare systems, and reimbursement frameworks across regions such as North America and Europe facilitate the translation of clinical advancements into precision medicine, enabling broader adoption of targeted conjugate therapies.

Logos and trademarks shown above are the property of their respective owners. Their use here is for informational and illustrative purposes only.
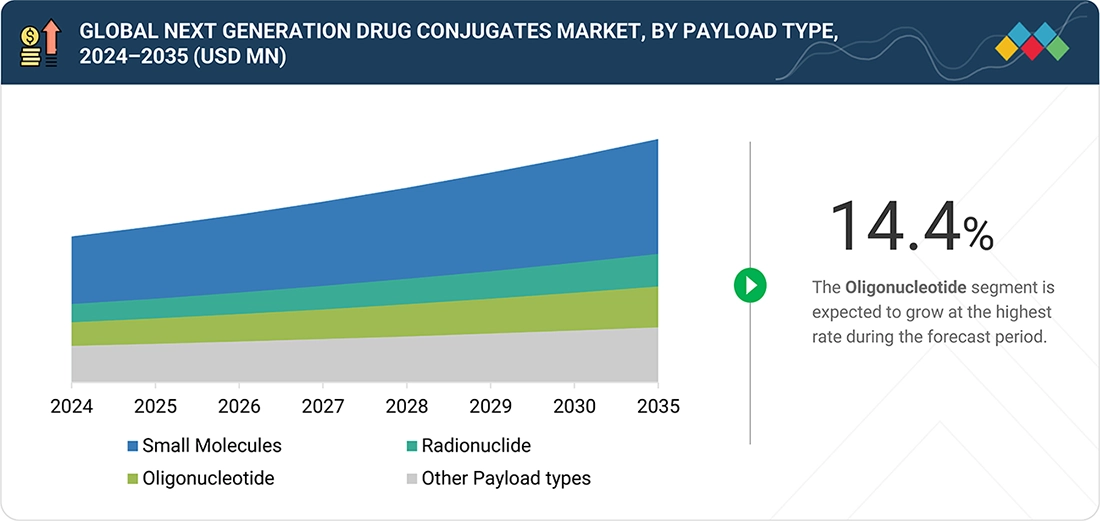
MARKET SEGMENTS
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
Global Next Generation Drug Conjugates Market, by Product
In 2025, Enhertu dominated the next generation drug conjugates market due to its strong clinical efficacy, expanding global approvals, and rapid adoption across HER2-targeted cancers. Its widespread use in breast cancer and growing indications in lung and gastric cancers have significantly contributed to its leadership.
Global Next Generation Drug Conjugates Market, by Type
In 2025, antibody–small molecule drug conjugates accounted for the largest share of the market, driven by their ability to combine monoclonal antibody specificity with highly potent cytotoxic payloads. This structure enables precise tumor targeting while minimizing systemic toxicity, making them the most commercially successful and clinically advanced class within next generation drug conjugates.
Global Next Generation Drug Conjugates Market, by Target Ligand
In 2025, antibody-based targeting ligands held the dominant share in the next generation drug conjugates market due to their high specificity toward tumor-associated antigens such as HER2. Their ability to selectively bind cancer cells and deliver payloads efficiently has positioned antibodies as the preferred targeting mechanism in most approved and pipeline ADCs
Global Next Generation Drug Conjugates Market, by Payload Type
In 2025, small molecule payloads dominated the market, owing to their high cytotoxic potency and effectiveness in killing cancer cells upon internalization. Payloads such as topoisomerase inhibitors and microtubule inhibitors are widely used in ADCs, enabling strong anti-tumor activity even at low doses while maintaining controlled toxicity profiles
Global Next Generation Drug Conjugates Market, by Indication
In 2025, breast cancer accounted for the largest share of the next generation drug conjugates market, primarily due to the high prevalence of the disease and the strong clinical success of HER2-targeted ADCs such as Enhertu. Continuous advancements in targeted therapies and expanding treatment eligibility for HER2-low populations have further reinforced breast cancer as the leading indication segment.
REGION
North America to be fastest-growing region in market during forecast period
During the forecast period, North America is expected to emerge as a leading and fast-growing region in the global NGDC market, driven by its strong ecosystem of biopharmaceutical companies, advanced research institutions, and specialized oncology centers. A robust pipeline of antibody-drug conjugates and other targeted conjugates, coupled with significant investments in precision medicine, biomarker-driven trials, and regulatory support, continues to accelerate market growth across oncology and expanding therapeutic areas.
NEXT GENERATION DRUG CONJUGATES (NDCS) MARKET: COMPANY EVALUATION MATRIX
In the global next generation drug conjugates (NGDC) market, AstraZeneca is the star player, driven by its strong portfolio of approved antibody-drug conjugates and a robust pipeline leveraging advanced linker and payload technologies. The company continues to lead innovation in targeted oncology therapies through strategic collaborations and sustained R&D investments. AbbVie is an emerging leader, gaining momentum through its expanding conjugate-based pipeline and partnerships, particularly in oncology and immunology. Its focus on next generation conjugation platforms and targeted delivery systems positions it strongly to capture future growth opportunities in the evolving NGDC landscape.
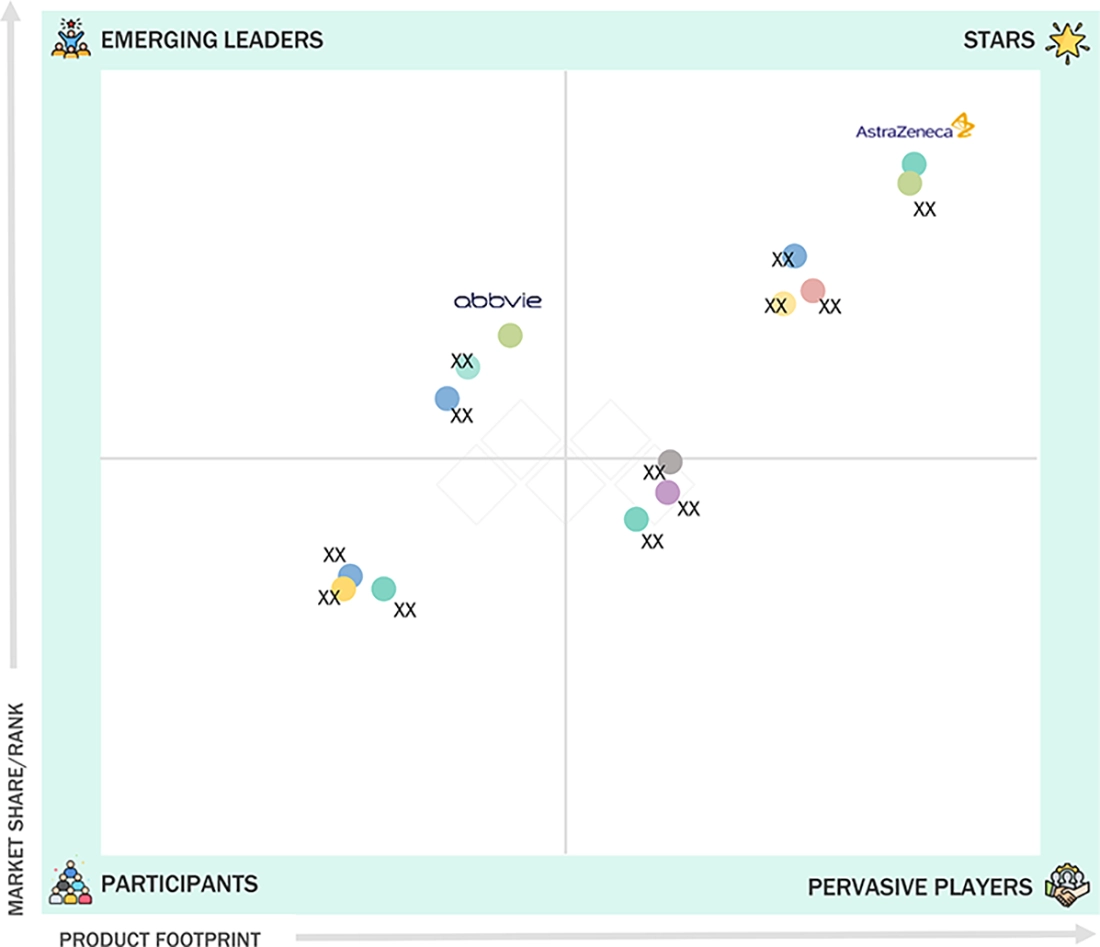
Source: Secondary Research, Interviews with Experts, MarketsandMarkets Analysis
KEY MARKET PLAYERS
- AstraZeneca (UK)
- Daiichi Sankyo, Company Limited (Japan)
- Novartis AG (Switzerland)
- Alnylam Pharmaceuticals, Inc (US)
- Gilead Sciences, Inc (US)
- AbbVie, Inc. (US)
- Ionis Pharmaceuticals, Inc. (US)
- Sanofi (France)
MARKET SCOPE
| REPORT METRIC | DETAILS |
|---|---|
| Market Size, 2025 (Value) | USD 13.94 Billion |
| Market Forecast, 2035 (Value) | USD 42.55 Billion |
| Growth Rate | CAGR of 11.7 % from 2026–2035 |
| Years Considered | 2024–2035 |
| Base Year | 2025 |
| Forecast Period | 2026–2035 |
| Units Considered | Value (USD Million/Billion) |
| Report Coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Segments Covered |
|
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
WHAT IS IN IT FOR YOU: NEXT GENERATION DRUG CONJUGATES (NDCS) MARKET REPORT CONTENT GUIDE
DELIVERED CUSTOMIZATIONS
We have successfully delivered the following deep-dive customizations:
| CLIENT REQUEST | CUSTOMIZATION DELIVERED | VALUE ADDS |
|---|---|---|
| Evaluating competitive positioning of NGDC pipelines | Analyzed pipelines of leading ADC and SMDC developers across clinical stages, target antigens, and payload innovations, benchmarking differentiation in efficacy, safety, and innovation focus | Support strategic decision-making by identifying whitespace opportunities, competitive gaps, and areas for pipeline expansion or licensing |
| Assessing high growth potential NGDC payloads | Supports strategic decision-making by identifying whitespace opportunities, competitive gaps, and areas for contract development and manufacturing service providers | Help evaluate key areas of entry in NGDC market for contract development and manufacturing service providers |
RECENT DEVELOPMENTS
- February 2025 : Alnylam collaborated with Regeneron to advance its RNAi therapeutic pipeline, including programs such as ALN-HTT02, targeting neurological and autoimmune diseases through RNA interference technologies.
- June 2025 : Gilead announced the acquisition of Interius BioTherapeutics to advance in-vivo cell therapy technologies aimed at improving targeted cancer treatment approaches and expanding its oncology innovation platforms.
Table of Contents

Methodology
This study involved four major activities in estimating the current size of the next generation drug conjugates market. Exhaustive secondary research was carried out to collect information on the market, its peer markets, and its parent market. These findings, assumptions, and sizing were then validated with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. Following this, the data triangulation procedure was used to estimate the market size of segments and subsegments.
Secondary Research
Secondary research was used mainly to identify and collect information for the extensive, technical, market-oriented, and commercial study. The secondary sources referred for this research study include publications from government sources, such as the National Center for Biotechnology Information (NCBI), National Institutes of Health (NIH), United States Food and Drug Administration (US FDA), European Medicines Agency (EMA), CT.gov, EudraCT, other clinical trial registries, News Articles, Press Releases, Paid Databases, Expert Interviews, and MarketsandMarkets Analysis, among others. Secondary sources also included corporate and regulatory filings (such as annual reports, SEC filings, investor presentations, and financial statements); business magazines and research journals; press releases; and trade, business, and professional associations. These sources were also used to obtain key information about major players, global product revenues, market classification, and segmentation according to industry trends, regional/country-level markets, market developments, and technology perspectives. Secondary data was collected and analyzed to arrive at the overall size of the global next generation drug conjugates market, which was validated through primary research.
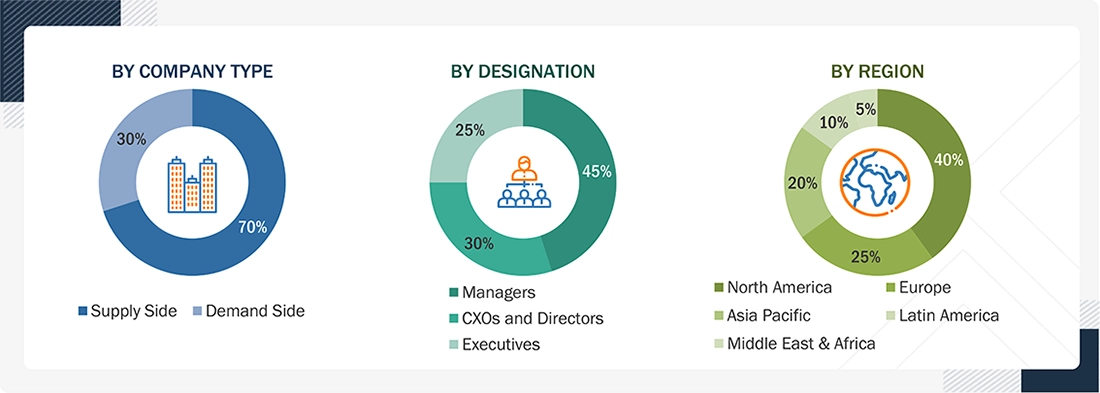
Primary Research
In-depth interviews were conducted with various primary respondents, including key industry participants, subject-matter experts (SMEs), C-level executives of key market players, and industry consultants, among other experts, to obtain and verify the critical qualitative and quantitative information as well as assess future prospects of the market. Various primary sources from both the supply and demand sides of the market were interviewed to obtain qualitative and quantitative information. The following is a breakdown of the primary respondents:
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
The global size of the next generation drug conjugates market was estimated through multiple approaches. A detailed market estimation approach was followed to estimate and validate the value of the market and other dependent submarkets. These methods were also used extensively to estimate the size of various subsegments in the market. The research methodology used to estimate the market size includes the following:

Data Triangulation
After estimating the overall market size from the market size estimation process, the total market was split into several segments. To complete the overall market engineering process and arrive at the exact statistics for all segments, the data triangulation procedure was employed; the data was triangulated by studying various factors and trends from both the demand and supply sides.
Market Definition
Drug conjugates are targeted therapeutic systems in which a biologically or chemically engineered targeting moiety (e.g., antibody, peptide, small-molecule ligand, or oligonucleotide delivery ligand) is conjugated to a therapeutic payload (e.g., cytotoxic drug, radionuclide, or nucleic acid) using linker or conjugation technologies. Next generation drug conjugates are defined by the use of advanced targeting platforms and/or novel payload technologies—particularly small molecules (e.g., topoisomerases, others), radionuclides, and oligonucleotides, enabled by improved linker and conjugation strategies, distinguishing them from earlier auristatin, maytansinoid, and calicheamicin-based conjugates. These enable improved precision, efficacy, and safety compared to earlier-generation conjugates.
Key Stakeholders
- Next generation drug conjugate manufacturing companies
- Distributors and suppliers of next generation drug conjugates
- Healthcare service providers (including hospitals & specialty clinics)
- National and regional research boards and organizations
- Research & development companies
- Clinical research organizations (CROs)
- Biotechnology companies
- Research laboratories & academic institutes
- Market research & consulting firms
- Regulatory bodies
Report Objectives
- To define, describe, and forecast the next generation drug conjugates market by product type, target ligand, payload type, indication, and region
- To provide detailed information regarding the major factors influencing the market growth (such as drivers, restraints, opportunities, and challenges)
- To analyze the micromarkets with respect to individual growth trends, prospects, and contributions to the overall next generation drug conjugates market
- To analyze the opportunities for stakeholders and provide details of the competitive landscape for market leaders
- To forecast the size of the market segments with respect to six regions: North America (US and Canada), Europe (Germany, France, the UK, Italy, Spain, and the Rest of Europe), Asia Pacific (China, Japan, India, South Korea, Australia, and the Rest of Asia Pacific), Latin America (Brazil, Mexico, Argentina, and the Rest of Latin America), the Middle East [GCC (Saudi Arabia (KSA), United Arab Emirates (UAE), and the Rest of GCC) and the Rest of Middle East], and Africa
- To profile the key players and analyze their market shares and core competencies
- To track and analyze competitive developments, such as product launches, partnerships, agreements, collaborations, and expansions
- To benchmark players within the market using the proprietary “Company Evaluation Matrix” framework, which analyzes market players on various parameters within the broad categories of business and product excellence strategy
Available customizations:
With the given market data, MarketsandMarkets offers customizations as per the company’s specific needs. The following customization options are available for the report:
Geographical Analysis
- Further breakdown of the Rest of Europe next generation drug conjugates market, by country
- Further breakdown of the Rest of Asia Pacific next generation drug conjugates market, by country
Company Information
- Detailed analysis and profiling of additional market players (up to five)
Segment Analysis
- Cross-segment analysis
Need a Tailored Report?
Customize this report to your needs
Get 10% FREE Customization
Customize This ReportPersonalize This Research
- Triangulate with your Own Data
- Get Data as per your Format and Definition
- Gain a Deeper Dive on a Specific Application, Geography, Customer or Competitor
- Any level of Personalization
Let Us Help You
- What are the Known and Unknown Adjacencies Impacting the Next Generation Drug Conjugates Market
- What will your New Revenue Sources be?
- Who will be your Top Customer; what will make them switch?
- Defend your Market Share or Win Competitors
- Get a Scorecard for Target Partners
Custom Market Research Services
We Will Customise The Research For You, In Case The Report Listed Above Does Not Meet With Your Requirements
Get 10% Free Customisation













Growth opportunities and latent adjacency in Next Generation Drug Conjugates Market