Clinical Trial Imaging Market by Software & Service (Operational Imaging, Trial Design), Modality (CT, MRI, X-Ray, PET, Ultrasound, ECHO), Therapeutic Area (Infectious, Oncology, CNS, CVD), End User (Pharma, Biotech, CROs) & Region - Global Forecast to 2028
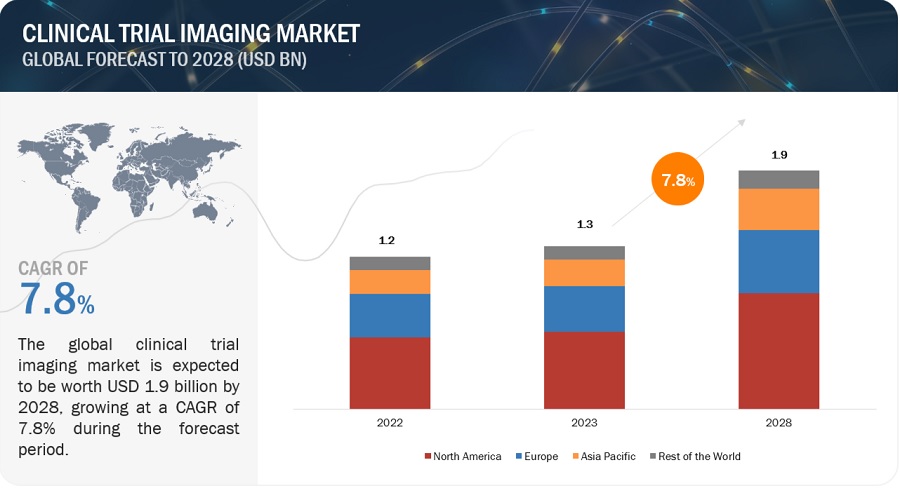
The global clinical trial imaging market in terms of revenue was estimated to be worth $1.3 billion in 2023 and is poised to reach $1.9 billion by 2028, growing at a CAGR of 7.8% from 2023 to 2028. The new research study consists of an industry trend analysis of the market. The new research study consists of industry trends, pricing analysis, patent analysis, conference and webinar materials, key stakeholders, and buying behaviour in the market. Increasing funding in research and development aimed at producing novel disease-fighting medications, coupled with the expansion of biotechnology and pharmaceutical firms, and increasing number of CROs, is expected to drive the market growth.
Attractive Opportunities in the Clinical Trial Imaging Market
To know about the assumptions considered for the study, Request for Free Sample Report

Clinical Trial Imaging Market Dynamics
Driver: Rise in research and development expenditures
Extensive investments in research for pioneering molecule development are a hallmark of pharmaceutical and biotechnology firms. The rise in both industry and government spending on research and development, combined with a growing emphasis on life science initiatives dedicated to creating novel therapeutic and diagnostic offerings, is projected to contribute positively to the global market.
Restraint: Significant expenses associated with the adoption of imaging systems
Within the healthcare sector, a substantial emphasis is placed on mitigating the escalating expenses associated with healthcare services. Numerous stakeholders in the industry are actively pursuing diverse strategies to achieve this objective. In this landscape, the challenge arises for providers of imaging services, as they grapple with the allocation of considerable capital to procure high-value assets like advanced radiology and other imaging systems. The expenses involved in deploying an imaging system encompass the acquisition, housing, and ongoing maintenance of the equipment. This initial financial burden, coupled with ongoing maintenance outlays, exerts a dampening influence on market expansion. Additionally, to effectively utilize such equipment, imaging service providers necessitate the engagement of skilled professionals, including experienced radiologists. These combined factors contribute to the substantial implementation costs associated with imaging systems, thereby acting as a significant deterrent to the growth trajectory of the global market
Opportunity: Growth opportunities in emerging economies
Emerging nations like India, South Korea, Brazil, and Mexico present substantial growth prospects for participants in the clinical trial imaging sector. This is primarily due to the escalating research and development investments within these countries. These developing markets also demonstrate a robust trend of translating life science research into commercial applications. Given the noteworthy advancements in life sciences research, these economies are poised to provide abundant growth avenues in the clinical trial imaging domain.
Furthermore, the Asian markets, particularly China and India, are home to numerous Contract Research Organizations (CROs) that furnish drug discovery services to pharmaceutical and biotechnology enterprises. With the marked upswing in research and development funding and the presence of CROs in emerging economies, the demand for clinical trial imaging is predicted to surge in these nations in the forthcoming years.
Challenge: Elevated expenses associated with conducting clinical trials
Research endeavors pertaining to healthcare progress invariably encompass clinical trials. These trials constitute a fundamental component for substantiating the efficacy of innovations, encompassing therapeutics, pharmaceuticals, medical apparatus, diagnostics, and scholarly investigations. While laboratory experiments strive to replicate human physiological conditions, the validation of a drug's effectiveness and safety within the targeted population necessitates a critical phase – conducting clinical trials. Nonetheless, orchestrating such trials to meticulously document the impact of therapies or drugs and vigilantly monitor potential adverse effects constitutes a financially demanding undertaking.
In 2022, services segment accounted for the largest share of the clinical trial imaging industry, by services and software
Based on product and services, the clinical trial imaging market is divided into services and software segments. Notably, in 2022, the services segment emerged as the dominant contributor to the market, primarily propelled by heightened research and development investments and the increase of the pharmaceutical and biotechnology sectors.
In 2022, computed tomography segment accounted for the largest share of the clinical trial imaging industry, by modality
The clinical trial imaging market is segmented into computed tomography, magnetic resonance imaging, ultrasound, positron emission tomography, X-ray, echocardiography and other modalities based on modality. Notably, in 2022, the computed tomography segment captured the predominant portion of the market share. This prominence can be attributed to factors including the rising presence of Contract Research Organizations (CROs), amplified research and development investments, and the expansion of the pharmaceutical and biotechnology sectors, collectively propelling this market segment.
In 2022, pharmaceutical companies segment accounted for the largest share in the clinical trial imaging industry, by end users
The clinical trial imaging market has been segmented into pharmaceutical companies, biotechnology companies, medical devices manufacturers, contract research organizations, academic and government research institutes and other end users based on end users. In 2022, the pharmaceutical companies segment accounted for the largest share of the market. The swift enlargement of this market is spurred by the escalating need for novel treatments and medications targeting chronic diseases. With the proliferation of pharmaceutical enterprises, manufacturers face intensified pressure to provide cutting-edge medications, striving to maintain competitiveness. The surge in demand for clinical trial imaging is projected to correlate with the remarkable array of innovative medicines unveiled by pharmaceutical companies, thereby playing a pivotal role in propelling the expansion of the market.
North America is the largest regional market for clinical trial imaging industry
The global clinical trial imaging market is segmented into five major regions—North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. In 2022, North America accounted for the largest share of the global market. The North American market's growth can be attributed to the, to the presence of big outsourcing companies and an increase in R&D in the region. Various factors such as the increasing geriatric population along with growing chronic diseases are projected to drive the market in North America. North America also performs the maximum number of clinical trials.
To know about the assumptions considered for the study, download the pdf brochure
The major players in clinical trial imaging market are ICON plc. (Ireland), BioTelemetry Inc. (US), Biomedical Systems Corporation (US), and Medpace Holdings, Inc. (US). These players lead the market because of their extensive product portfolios and wide geographic presence. These dominant market players also have numerous advantages, such as more robust marketing and distribution networks, larger budgets for R&D, and better brand recognition.
Scope of the Clinical Trial Imaging Industry
|
Report Metric |
Details |
|
Market Revenue in 2023 |
$1.3 billion |
|
Projected Revenue by 2028 |
$1.9 billion |
|
Revenue Rate |
Poised to Grow at a CAGR of 7.8% |
|
Market Driver |
Rise in research and development expenditures |
|
Market Opportunity |
Growth opportunities in emerging economies |
This report categorizes the clinical trial imaging market to forecast revenue and analyze trends in each of the following submarkets:
Service and Software
-
Services
- Operational Imaging Services
- Read Analysis Services
- System and Technical Support Services
- Trial Design & Consulting Services
- Software
By Modality
- Computed Tomography
- Magnetic Resonance Imaging
- Ultrasound
- Positron Emission Tomography
- X-ray
- Echocardiography
By Therapeutic area
- Oncology
- Infectious Diseases
- Neurology
- CVS
- Endocrinology
- Immunological Disorder
By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Manufacturer
- Contract Research Organizations
- Academic and Government Research Institutes
By Region
-
North America
- US
- Canada
-
Europe
- Germany
- UK
- France
- Italy
- Spain
- Rest of Europe
-
Asia Pacific
- China
- Japan
- India
- Rest of Asia Pacific
- Latin America
- Middle East & Africa
Recent Developments of Clinical Trial Imaging Industry
- In March 2023, ICON Plc. (Ireland) announced a strategic partnership with LEO Pharma (Denmark) to scale clinical trial execution that is patient-centric and cost effective, and which will support the company’s overall ambition of building one of the most effective and efficient clinical portfolio execution organisations in the industry.
- In March 2023, Clario (US), announced the launch of a cutting-edge cloud-based image viewer tool for sponsors and contract research organizations to view images related to their clinical trials.
- In May 2021 IXICO Plc. (UK) signed a contract with biopharmaceutical client IXICO to provide neuroimaging services for a Phase III clinical trial under this contract.
Frequently Asked Questions (FAQ):
What is the projected market revenue value of the global clinical trial imaging market?
The global clinical trial imaging market boasts a total revenue value of $1.9 billion by 2028.
What is the estimated growth rate (CAGR) of the global clinical trial imaging market?
The global clinical trial imaging market has an estimated compound annual growth rate (CAGR) of 7.8% and a revenue size in the region of $1.3 billion in 2023.
To speak to our analyst for a discussion on the above findings, click Speak to Analyst
The objective of the study is to analyze the key market dynamics, such as drivers, opportunities, challenges, restraints, and key player strategies. To track company developments such as acquisitions, product launches, expansions, collaborations, agreements, and partnerships of the leading players, the competitive landscape of the clinical trial imaging market to analyze market players on various parameters within the broad categories of business and product strategy. Top-down and bottom-up approaches were used to estimate the market size. To estimate the market size of segments and subsegments, market breakdown and data triangulation were used.
The four steps involved in estimating the market size are:
Collecting Secondary Data
The secondary research data collection process involves the usage of secondary sources, directories, databases (such as Bloomberg Businessweek, Factiva, and D&B), annual reports, investor presentations, and SEC filings of companies. Secondary research was used to identify and collect information useful for the extensive, technical, market-oriented, and commercial study of the market. A database of the key industry leaders was also prepared using secondary research.
Collecting Primary Data
The primary research data was conducted after acquiring knowledge about the market scenario through secondary research. A significant number of primary interviews were conducted with stakeholders from both the demand side (such as hospitals & ICUs, diagnostic laboratories, research institutes, research laboratories and point-of-care settings) and supply side (such as included various industry experts, such as Directors, Chief X Officers (CXOs), Vice Presidents (VPs) from business development, marketing and product development teams, product manufacturers, wholesalers, channel partners, and distributors) across major countries of North America, Europe, Asia Pacific, the Middle East & Africa, and Latin America. Approximately 40% of the primary interviews were conducted with stakeholders from the demand side, while those from the supply side accounted for the remaining 60%. Primary data for this report was collected through questionnaires, emails, and telephonic interviews.
A breakdown of the primary respondents is provided below:
The following is a breakdown of the primary respondents:
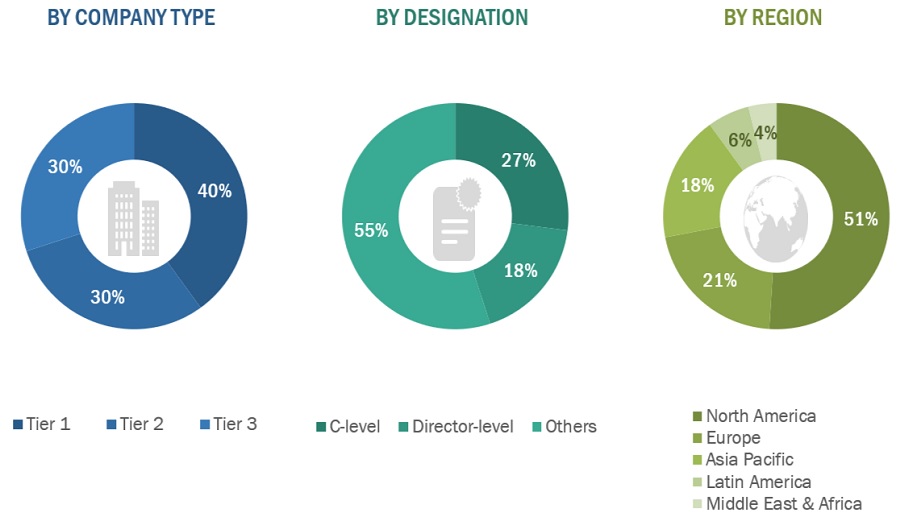
Note 1: *Others include sales managers, marketing managers, and product managers.
Note 2: Tiers are defined based on a company’s total revenue as of 2022: Tier 1=> USD 1 billion, Tier 2 = USD 500 million to USD 1 billion, and Tier 3=< USD 500 million.
To know about the assumptions considered for the study, download the pdf brochure
Market Size Estimation
All major product manufacturers offering various clinical trial imaging products were identified at the global/regional level. Revenue mapping was done for the major players and was extrapolated to arrive at the global market value of each type of segment. The market value clinical trial imaging market was also split into various segments and subsegments at the region and country level based on:
- Product mapping of various manufacturers for each type of market at the regional and country-level
- Relative adoption pattern of each market among key application segments at the regional and/or country-level
- Detailed primary research to gather qualitative and quantitative information related to segments and subsegments at the regional and/or country-level.
- Detailed secondary research to gauge the prevailing market trends at the regional and/or country-level
Global Clinical Trial Imaging Market Size: Top-Down Approach

To know about the assumptions considered for the study, Request for Free Sample Report
Data Triangulation
After arriving at the overall market size—using the market size estimation processes—the market was split into several segments and subsegments. To complete the overall market engineering process and arrive at the exact statistics of each market segment and subsegment, the data triangulation, and market breakdown procedures were employed, wherever applicable. The data was triangulated by studying various factors and trends from both the demand and supply sides in the clinical trial imaging industry.
Market Definition
Clinical trial Imaging is used to create images during clinical trials while conducting research. Such imaging testing is mainly done using X-rays, computed tomography (CT), magnetic resonance imaging (MRI), ultrasound, and positron emission tomography (PET). In clinical trials, imaging is used primarily to evaluate efficacy, endpoints, and more frequently, for safety evaluations and eligibility criteria.
Key Stakeholders
- Medical Device Industries
- Academic and Government Research Institutes
- Diagnostic Centers
- Market Research & Consulting Firms
- Clinical Trial Imaging Manufacturers, Vendors, and Distributors
- Pharmaceutical and Biotechnology Companies and Contract Research Organizations (CROs)
Report Objectives
- To define, describe, segment, and forecast the clinical trial imaging market by service & software, modality, therapeutic area, end user, and region
- To provide detailed information regarding the major factors influencing the market growth (drivers, restraints, opportunities, and challenges)
- To analyze micromarkets1 with respect to individual growth trends, prospects, and contributions to the overall market
- To analyze market opportunities for stakeholders and provide details of the competitive landscape for key players
- To forecast the size of the market segments with respect to five regions, namely, North America, Europe, the Asia Pacific, Latin America, and the Middle East & Africa
- To profile the key players and comprehensively analyze their product portfolios, market positions, and core competencies
- To track and analyze company developments such as acquisitions, agreements, partnerships, contracts, approvals, product launches, and mergers in the clinical trial imaging market
- To benchmark players within the market using the proprietary “Company Evaluation Matrix” framework, which analyzes market players on various parameters within the broad categories of business and product strategy
Available Customizations
With the given market data, MarketsandMarkets offers customizations as per the company’s specific needs. The following customization options are available for this report:
Country Information
Clinical trial imaging market size and growth rate estimates for countries in the Rest of Europe, the Rest of Asia Pacific, Latin America, and Middle East & Africa.
Company profiles
An additional fifteen company profiles of players operating in the clinical trial imaging market.



 Generating Response ...
Generating Response ...







Growth opportunities and latent adjacency in Clinical Trial Imaging Market
Decode Al development designed nano human anology of all engineering military weaponry development